Recruiting Black volunteers for COVID-19 vaccine trials means overcoming mistrust
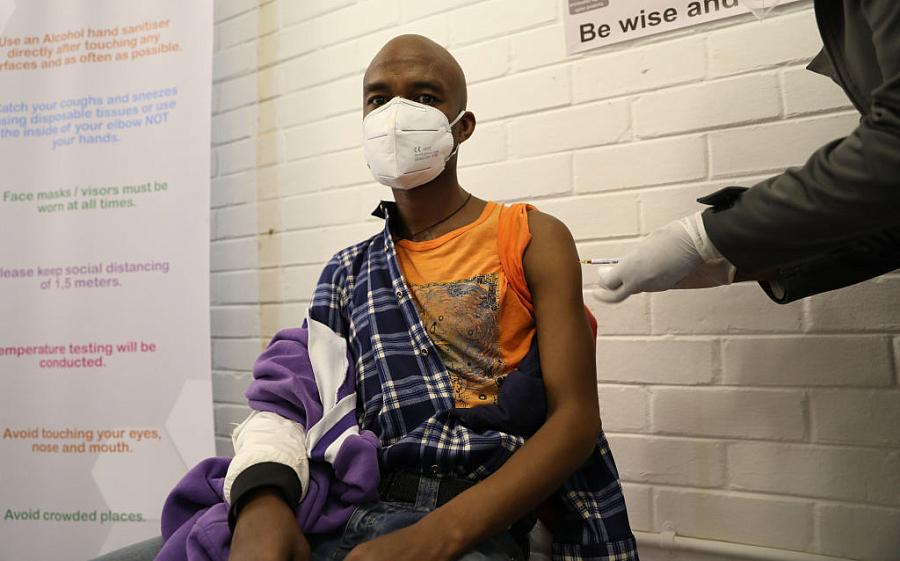
A volunteer participating in Oxford’s COVID-19 vaccine trial receives a shot in South Africa on June 24.
(Photo by Siphiwe Sibeko/POOL/AFP/Getty Images)
A year ago the word corona was associated with beer. And now look at where we have landed. Who would have thought we would wake up every day hoping that science will deliver us from this COVID nightmare? It can happen, but it’s going to take some serious experimentation and patience to get us there. That means someone — or many people — will have to participate in clinical trials.
Recruitment and participation in clinical trials is a sensitive subject for racial minorities. Can a safe, effective, desperately needed vaccine be created without our participation in research? Why is it so important that Black and Brown people donate their time and anatomy to science? Although the coronavirus has wrecked so many lives and trashed our economy, these questions are loaded and not easy to answer.
Recruiting volunteers
In a perfect world, scientists would recruit volunteers from several communities that represent a diverse population to participate in clinical trials. They would make a special effort to get volunteers from populations disproportionately affected by the targeted illness. Unfortunately, not everyone will eagerly volunteer.
Blacks make up about 14% of total clinical trial participants in the United States, roughly proportionate to their representation in the population, though they are at higher risk of many serious illnesses. In clinical trials for cancer treatments, for example, participation of Black people runs low, though they have a higher death rate from cancer. According to a ProPublica analysis, Black participants accounted for fewer than 5% of patientswho took part in the studies that produced 24 of the 31 cancer drugs approved by theFood and Drug Administration over the last five years.
Moderna, the Cambridge, Massachusetts, biotech company considered one of the front-runners in the race to develop a COVID-19 vaccine, recently released data on the early-stage trial. Of the 45 healthy volunteers, 13% were Hispanic, 4% were Black, and only 2% were Asian.
There is sufficient evidence that the coronavirus is hitting the African American community harder than others. Blacks and Hispanics have a greater chance of exposure for a number of reasons, including employment that is not remote-friendly and higher rates of underlying medical conditions. This suggests that nowhere is a vaccine of greater importance than in the Black community.
But skepticism is a bitter pill to swallow.
As a health journalist I mingle in different circles to see what people think about trending health topics. I spoke with a few health professionals about the possibility of participating in a clinical trial for a COVID-19 vaccine.
“It would be extremely difficult to participate in a vaccine trial for COVID-19,” said Sharon Evans, an independent public health consultant in Raleigh, North Carolina. “The main reasons are there has been a lack of authentic transparency when dealing with this issue, lack of testing, inconsistent protocols, constant changes in information that is being disseminated.”
Deidra Sorrell, a licensed clinical professional counselor in Waldorf, Maryland, rejects the notion of participating in clinical trials while questioning the origins of the virus. “I realize that creating a safe vaccine requires a diverse group of participants for trials but I have an extreme distrust of anything associated with COVID-19. The origin of this virus is very sketchy in my opinion.”
I spoke to Amer-Marie Woods, an African American neurophysiologist in Atlanta, about the lack of trust she has observed with her patients. “I have had patients that refuse to talk to the (White) doctor but will talk to me. That shows that we need to see more Black doctors in these spaces.”
Historical scars linger
Whenever I hear discussions among Black Americans about clinical trials, there's a collective groan of disapproval. Everyone knows how the medical community has betrayed our confidence through clandestine experimentation. When researchers recruit in the Black community, a wall comes up.
I spoke with a friend recently about COVID-19 trials. She worked to help recruit people from “underserved” and “disadvantaged” populations into clinical trials until she did a little research. Melva Florence, executive director of The LaStraw Inc., a community nonprofit in Greensboro, North Carolina, said she is now wary of talking people into volunteering for experimentation.
“When we are out here encouraging people to participate in vaccine trials, we have no way of assuring them that they will not be victimized,” she said. “We also can’t in good conscience dismiss their concerns. The more I learned, the more foolish I felt. I actually felt like a foil. My Black face was being used to earn a trust in the medical field that they didn’t earn and do not deserve.”
I reflect on the abuses committed by J. Marion Simms, the so-called “father of modern gynecology.” His claim to fame was developing surgical methods in the mid-1800s, such as gallbladder surgery and artificial insemination. But his experiments were performed on enslaved Black women who were not anesthetized. That painful reputation has carried on throughout the years.
There’s the infamous Tuskegee Experiment in the fall of 1932, in which Black men in the rural South were told they were being treated for “bad blood.” In reality, scientists used them as human subjects to see what would happen when syphilis was left untreated.
Then there is Henrietta Lacks, an African American woman in Baltimore who suffered from advanced cervical cancer. She died in 1951, but not before a White surgeon took samples from her tumors and used them to study human cell perpetuity. Her cells are responsible for developing many drugs and vaccines including the one that, ironically, is used to prevent cervical cancer.
I also cringe as I think about the Eugenics Board of North Carolina. From 1929 to 1974, this state-approved board sterilized more than 7,600 people by force or coercion. They were mostly poor, Black young women who were victims of rape or incest, or born with a mental or physical disability. It’s only recently that these victims are being compensated, but not all of them will see the restitution they are due.
In addition, there’s a continuous and contentious debate about using people in African countries to test vaccines. Many nations are still scarred from decades of experimentation. Recently, French researchers suggested that COVID-19 drug trials begin in Africa. The idea was rebuffed with the hashtag #Africansarenotguineapigs.
If there is any question about the intentions of medical researchers, it is nearly impossible to suppress the communal anxiety that will surely grow.
Why is it important to have racial minorities involved in clinical studies?
On the other side of this debate is the integrity of scientific inquiry. Will a vaccine tested on White bodies react the same in other bodies? How can researchers reduce or eliminate the fear of science enough to recruit Black and Brown people to test a potentially life-saving vaccine?
Black and Hispanic representation in clinical trials are traditionally low because of barriers such as mistrust, low health literacy, cost of transportation, child care, and possible missed work days. But homogeneous testing is discouraged in trials. In many cases patients of color will ultimately take the drugs developed in trials, so keeping them involved in the process is critical. Drugs and vaccines may not react the same among all ages, races and ethnicities.
Florencia Gonzalez, Community Networks Manager at Georgetown-Howard Universities Center for Clinical and Translational Science (GHUCCTS), said it isn’t normally good practice to apply test results so broadly. “Historically, people of color are underrepresented in clinical trials. I do feel that doctors are doing their best to include more minorities but it takes time. Unfortunately, there has not been any systematic tracking or study to ensure that diverse communities are being included.”
The Food and Drug Administration has issued guidance and started an initiative on how to recruit more minority populations in trials.
So, where do we go from here? Carla Williams, associate professor of medicine and public health at Howard University and co-director for community engagement at GHUCCTS, says safety is a priority. “People have to feel safe, included and respected. Safety is always top of mind for both researchers and participants. It’s important that people know that there are strong safeguards in place to protect their safety and confidentiality. This includes a careful process of reviewing a research project before giving approval to start enrolling people in a study.”
Gonzalez adds that creating videos, having translators on hand, and simplifying educational materials can lower the stress of trying to make a breakthrough. “For people with limited English proficiency, we’re trying to ensure there is quality language and interpreters available to explain things clearly. We make sure that consent forms are translated. We try to consider the health literacy levels when creating educational materials.”
GHUCCTS has recently published a blog post and a short video about the importance of getting Black and Hispanic people on board with clinical trials for COVID-19. For additional resources, check out the clinical trials department of Keck School of Medicine of the University of Southern California.

